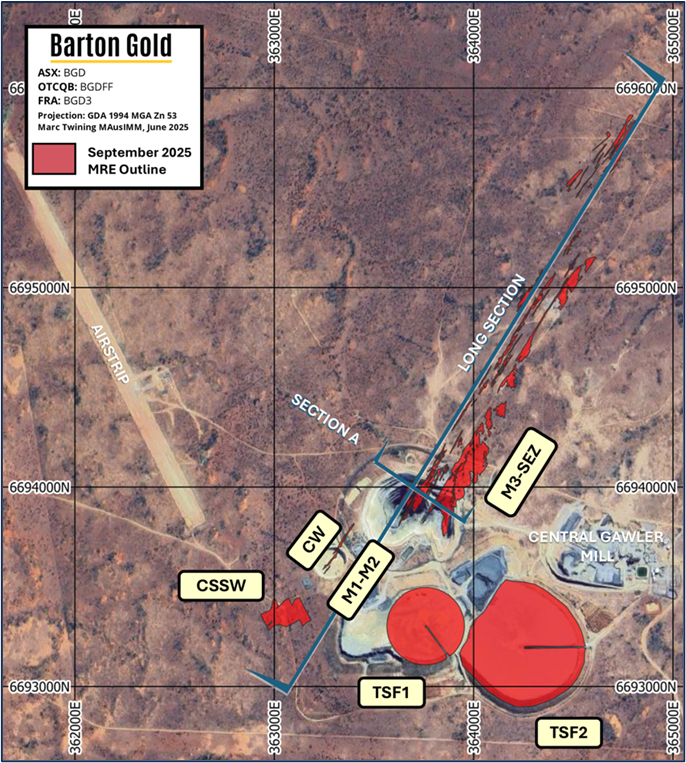
ISO 17025-accredited California lab offers HPLC, LC-MS/MS, ICP-MS, and ICH stability studies with 48-hour turnaround for FDA submissions.
IRVINE, CA, UNITED STATES, March 17, 2026 /EINPresswire.com/ — Qalitex Laboratories, an ISO 17025-accredited third-party analytical testing laboratory serving pharmaceutical developers, contract research organizations (CROs), and dietary supplement brands across North America, today highlighted the pharmaceutical testing requirements that drug developers and small biotechs must address when preparing IND and NDA submissions to the FDA — and how analytical chemistry testing, stability studies, and method validation integrate into the regulatory timeline.
FDA requirements for pharmaceutical analytical testing are governed by ICH guidelines, which define standards for stability testing, method validation, and impurity profiling. ICH Q1A(R2) establishes stability testing requirements for new drug substances and products, including storage conditions, testing intervals, and the data package required for regulatory submissions. ICH Q2(R1) defines validation requirements for analytical procedures, including specificity, linearity, range, accuracy, precision, and detection and quantitation limits.
For drug developers preparing IND filings, stability data is a mandatory component of the chemistry, manufacturing, and controls (CMC) section. FDA requires data demonstrating that the drug substance and product maintain acceptable quality throughout the proposed shelf life. For early-stage IND submissions, accelerated stability data at 40°C / 75% RH per ICH Q1A(R2) is typically required, with long-term stability studies at 25°C / 60% RH initiated at the time of filing.
“The challenge for small drug developers and biotechs is that large CROs often have 6-to-12-week wait times for routine analytical testing and stability program initiation,” said Nour Abochama, Vice President of Operations at Qalitex Laboratories. “When you’re working toward an IND filing deadline, that timeline can be a significant constraint. We built our pharmaceutical testing program to provide CRO-quality analytical results — validated methods, ICH-compliant stability chambers, pharmaceutical-grade QC documentation — with turnaround times that align with real development schedules.”
Qalitex’s pharmaceutical analytical testing capabilities include:
HPLC and UPLC for identity, potency, and purity
LC-MS/MS for impurity profiling and trace-level quantitation
ICP-MS for elemental impurity analysis per ICH Q3D
GC-MS for residual solvent testing per USP and ICH Q3C
Dissolution testing per USP
Content uniformity per USP
All methods are validated per ICH Q2(R1) or developed against applicable USP monographs.
Stability testing at Qalitex is conducted in ICH-qualified chambers with continuous temperature and humidity monitoring and automated data logging. The laboratory offers long-term stability at 25°C / 60% RH (ICH Zone II), intermediate at 30°C / 65% RH, and accelerated at 40°C / 75% RH, with testing at ICH-specified time points of 0, 3, 6, 9, 12, 18, 24, and 36 months. Photostability testing per ICH Q1B is also available.
California’s life sciences sector includes more than 3,000 companies, with a significant concentration of small and mid-size drug developers and biotechs in the San Diego and Los Angeles corridors. Many lack in-house analytical testing infrastructure and rely on contract testing laboratories for both development-stage and commercial-stage analytical work.
“The questions that matter most when a pharmaceutical developer evaluates a testing laboratory are about method validation and regulatory defensibility,” said Abochama. “Is the method validated for your specific matrix? What reference standards were used? Is the stability chamber ICH-qualified with continuous monitoring? Can you provide the validation data for inclusion in your regulatory submission? We answer these questions directly, and the documentation we provide is designed to be submission-ready.”
All pharmaceutical testing at Qalitex is performed under FDA 21 CFR Part 211 cGMP guidelines alongside ISO 17025 accreditation standards.
Resources:
Full article: https://qalitex.com/services/pharmaceutical-testing/
About Qalitex Laboratories
Qalitex Laboratories is an ISO 17025-accredited third-party analytical testing laboratory with facilities in Irvine and San Diego, California. The laboratory provides certificate of analysis (COA) testing, heavy metal analysis by ICP-MS, microbiology testing per USP and , preservative efficacy testing, stability studies under ICH guidelines, and regulatory compliance support for dietary supplement brands, cosmetic companies, and consumer goods manufacturers. Turnaround times start at 48 hours for standard panels. Testing programs meet 21 CFR Part 111, California Proposition 65, Amazon supplement compliance requirements, and Health Canada NHP Directorate standards.
Nour Abochama
Qalitex Laboratories
+1 949-881-6661
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
Legal Disclaimer:
EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
![]()